Introduction
Sterilization is the process of eliminating and controlling bacteria or other living microorganisms on the surface of an article or fluid to avoid the transmission of these microorganism which have potential to make threat to the patient’s life. All-important medical devices that come into touch with sterile tissues, as well as "semi-critical" devices that encounter mucous tissues or non-intact skin, must be sterilized. There are different kinds of sterilization methods Gamma Sterilization, ETO Sterilization, vapor hydrogen peroxide sterilization (Hybrid), Electron Beam Sterilization, X Ray Sterilization, Steam Sterilization are some of the examples. Sterilization processes are controlled and standardized by FDA (Food and Drug Administration), ISO.
Why Sterilization Is Important
A surgical equipment or medical device that encounters a patient's mucous membranes or sterile tissue and poses a considerable risk of introducing dangerous germs and infection. Because of the breach of host barriers, improper sterilization and disinfection of medical equipment raises the risk of infection. Getting rid of blood, mucus, saliva, pus, foreign particles, or debris that could cause serious complications for the next patient who needs surgery, and the medical practitioner utilizes the equipment so it’s essential to do sterilization of Packaging to avoid threat to patients’ life.
Different Types of Sterilization Methods
Gamma Sterilization - Gamma rays which are emitted by cobalt-60 radiation source is a vital part of sterilization process. Gamma rays’ sterilization is an irradiation process where the product is exposed to gamma rays it is one of the common methods of sterilization process because it is predictable, simple only need low skill, and with minimum line of maintenances it can be done n number of time without any reduction in accuracy. In gamma ray sterilization the gamma rays are emitted from a radioactive isotope to the product in order to kill all the microbes. These radiations have the ability to penetrate through the package and packaging as electromagnetic wave so the product can be sterilized without opening seal of the packaging, these will result in faster sterilization and shipment.
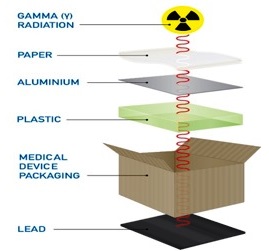
ETO (Ethylene oxide Sterilization) - It is the process in which the product is exposed to the gas at a specific temperature, pressure, and humidity for a controlled period. It can used to all most all kind of material even for polymers without changing the physical chemical and biological structures and these processes can be done in house. These gases are hazardous to human(carcinogen) and environment if not used carefully can be exploded.
Hybrid Method - Vapor Hydrogen peroxide method: it is a low temperature sterilization process .it can used as physical method and chemical method. These processes are more viable for heat sensitive products and only consumes less time than others. These sterilization is done by filling the vapor of H2O2 in a chamber and exposing the package and packaging into it . H2O2 sterilization is one of the best environmentally friendly method as it doesn’t have any hazardous residue, the only residue are water and oxygen so it doesn’t require any ventilation. It is very low cost method and can be performed in-house. Even though it cannot handle cellulosic materials and don’t have high penetration.

Electron Beam Sterilization - Electron beam accelerator are used to carry out this sterilization method in this method using a high power electron beam accelerator a powerful electron beam is created. The beam is swept back and forth to form to form curtain of rapid electrons that rain and ionize the materials they come into contact within electron beam sterilization, single-layer boxes containing medical devices are placed on a conveyor. Electrons penetrate the cardboard box and all the medical instruments in their respective containers inside the carton as they pass through the beam. Harmful germs are fully destroyed with minimal impact on medical equipment. The radiation dose decreases as the electrons penetrate the products thus less radiation exits the box than enters. To achieve a generally consistent dose, medical instruments are usually turned over and irradiated from the opposite side. There is no radioactivity in this method.
X Ray Sterilization - This method is a irradiation process using the photon radiation. X-ray sterilization, like gamma and E-Beam sterilization, employs irradiation to destroy the DNA of microbes on the product. X-rays are electromagnetic photons with high energy, high frequency, and short wavelength. X-rays have deeper penetration and allows to use in all kinds of material like liquids, metals etc. X-rays provide uniformity in dose and provides tighter dosed range, It can be used for sterilization of polymers which are reactive to heat and chemicals as it only have low impact on polymers .X ray also have fastest processing speed with less impact on nature.
Steam Sterilization – Steam sterilization is nontoxic, low cost, microbicidal and sporicidal, warms and penetrates textiles quickly. The basic idea of steam sterilization in an autoclave is to expose each item to direct steam contact for the prescribed period at the requisite temperature and pressure. Steam sterilization uses four parameters: steam, pressure, temperature, and time. Dry saturated steam with entrained water (dryness fraction 97%) is the best steam for sterilizing. Pressure is used to achieve the high temperatures required to destroy bacteria fast. To achieve microbicidal efficacy, certain temperatures must be reached. 121°C (250°F) and 132°C (270°F) are the two most prevalent steam-sterilizing temperatures. To kill microbes, these (and other high temperatures)830 must be sustained for a short period of time. 30 minutes is the recognized minimum exposure length for sterilizing wrapped healthcare products.
Flash sterilization - Flash sterilization is a variation on traditional steam sterilization (gravity, pre-vacuum, or steam-flush pressure-pulse) in which the flashed object is put in an open tray or a specifically constructed, covered, rigid container to allow for quick steam penetration. Because of the lack of timely biological indicators to monitor performance, the absence of protective packaging following sterilization, the possibility of contamination of processed items during transportation to operating rooms, and the sterilization cycle parameters (i.e., time, temperature, pressure) are minimal, it is not recommended as a routine sterilization method.
Peracetic acid sterilization - Peracetic acid is a highly biocidal oxidant that retains its effectiveness even when organic soil is present. Surface pollutants are removed with peracetic acid. A single-dose container contains the sterilant, 35 percent peracetic acid, and an anticorrosive agent. The container is perforated at the point of use, just before the lid is closed and the cycle begins. At a temperature of around 50°C, concentrated peracetic acid is diluted to 0.2 percent with filtered water (0.2 mm). For 12 minutes, diluted peracetic acid is cycled within the machine's chamber and pushed through the endoscope's channels, decontaminating external surfaces, lumens, and attachments.
Dry heat sterilization - This approach should only be used on materials that are susceptible to moisture damage or are resistive to moisture. Dry heat offers the following advantages: it is harmless and does not affect the environment; a dry heat cabinet is simple to build and operate. It penetrates materials; and it is noncorrosive to metal and sharp equipment. Static-air sterilizers and forced-air sterilizers are the two types of dry-heat sterilizers. The oven-type sterilizer is named after the heating coils in the bottom of the machine, which induce hot air to ascend within the chamber by gravity convection. Dry-heat sterilizers are slower to heat, take longer to achieve sterilizing temperatures, and have less consistent temperature control throughout the chamber than forced-air sterilizers. A motor-driven blower pumps heated air around the chamber at a high velocity in a forced-air or mechanical convection sterilizer, allowing for a faster transfer of energy from the air to the instruments.

Chemical Sterilization- Chemicals can also be used to sterilize samples that cannot be sterilized with heat, such as biological samples, polymers, and electronic components. The organisms are exposed to a high concentration of a reactive gas such as ozone, nitrogen dioxide, or alkylating chemicals in gas sterilizers. Chlorine, formaldehyde, glutaraldehyde, quaternary ammonium salts, and ETO are the most often utilized compounds for this. However, because of their limited capacity to penetrate bacterial spores, they are mostly used to sterilize medical equipment rather than biological substances, posing a health risk to the operators.
Factors to be Considered while Choosing Sterilization Method
• Understanding the difference of each sterilization and how they affect packaging material -There are many kinds of sterilization each of them are entirely different by other so we have to consider the packaging and package material before selecting sterilization method , for e.g. some products such as polymer are sensitive to heat so for such products auto clave methods are not suitable so the sterilization methods like ETO are chosen , likewise some material are sensitive to radiation so it is necessary to understand the difference in each methods
• Time associated with each cycle - it is necessary to take in to account the time associated with each sterilization methods. Some sterilization methods are very time consuming the time consumption will cause less productivity and will result in loss, so it is very important to use the alternative sterilization methods. For e.g., ETO sterilization is more time consuming than the HYBRID method so in some instance it can be used to avoid the time consumption.
• Use of vendors versus in-house sterilization-Many sterilization methods are expensive so there are limitations for the company to use such sterilization methods so they outsource this process to a contracting company, while selecting a sterilization process it is mandatory to check whether it can be performed in-house.
• Overall cost associated with each methods-It is essential to consider the cost associated with each method and have to choose the cost effective and qualified methods for example the cost of X-RAY sterilization, Gamma ray sterilization, Electron beam sterilization are low to compared to other kind of methods so it can be substituted to the expensive methods this will result in reduction in Cost and increase in turnover
FDA STERILIZATION PROCESS CONTROLS
Inspectional Objectives
1. Confirm that the sterilization process was validated by reviewing the validation study. Validation studies are necessary for the sterilization processes. Validation study assessment includes and not only limited to review of validation study summary and approval if the process is already established and previously assessed.
2. Review the specific procedure(s) for the sterilization process selected and the methods for controlling and monitoring the process. Verify that the process is controlled and monitored. *All the procedures used in the sterilization procedure must be controlled and monitored with great care. Ensuring that the sterilization process adhere to the process followed in the standard documents such as Device Master Record (DMR) is also a key pointer.
3. If review of the Device History Records (including process control and monitoring records, acceptance activity records, etc.) reveals that the sterilization process is outside the firm's tolerance for operating or performance parameters:
a) Determine whether the nonconformance’s were handled appropriately; and
b) Review the equipment adjustment, calibration, and maintenance*
4. If the sterilization process is software controlled, confirm that the software was validated.
It is required to validate all the software’s used in the process of sterilization.
Reviewing the software requirements document, software update history, software validation protocol, software validation activities, software change controls and software validation results are the key pointers which should be considered with great importance.
5. Verify that personnel have been appropriately qualified and trained to implement the sterilization process. Inspecting and verifying the personnel working in the sterilization process are well qualified and well trained to implement the roles and duties assigned to them is very important. Sterilization process is a complicated process hence it should be undertaken by qualified and trained personnel only.
ISO Standard ( International Organization for Standardization)

ISO is a certification that demonstrates that a manufacturing or service business adheres to the International Organization for Standardization (ISO) requirements, which include strong management, product development documentation, and excellent customer service procedures, quality etc.
The ISO sterilization standards publications are separated into two parts: a requirement portion and a guideline section The material in the prerequisites section provides fundamental and required provisions for the proper execution of the procedures indicated. These requirements should be included into the manufacturer's procedures, and technical information reports (TIR) are extra documentation that assist the manufacturer in meeting the requirements
Following are examples of some ISO standards
ISO 11135:2014 establishes criteria for the development, validation, and routine control of an ethylene oxide sterilization process for medical devices in both industrial and health-care environments, taking into account the similarities and variations between the two applications.
ISO 11137: Radiation sterilization-This section of ISO 11137 covers techniques for determining the sterilizing dosage using one of the two procedures described in ISO 11137-1:2006.These approaches employ the following methods:
- Dosage setting to acquire a product-specific dose
- Dose substantiation to confirm a preselected dose of 25 kGy or 15 kGy.
ISO 20857:2010: standards for the development, validation, and routine control of a dry heat sterilization process for medical devices It primarily focuses on dry heat sterilization, but it also includes standards and guidelines for dehydrogenation techniques that use dry heat
ISO 22441: Sterilization Requirements for the development, validation, and routine control of a sterilization process for medical devices or health care products using Low temperature vaporized hydrogen peroxide.
Packaging and Sterilization
In the development of safe, effective, and regulatory-compliant medical devices, packaging and sterilization are critical. Sterilization guarantees that a medical equipment is free of microorganisms, whereas packaging protects a medical product during transport or storage and aids in aseptic presentation to end users. To ensure the aseptic presentation and integrity of Packaging various test are conducted. Testing the integrity of packaging seals help to ensure there is required amount of protection. Seal integrity is the most important method to maintain the quality of a product. If there is no seal integrity test, there is chance of risk of breach in the seal and can cause damage to the sterility and quality of the product.
A good quality package seal can protect the product from dust, wrinkles, and other physical, chemical, and biological particles, to make sure the package seals are competitive package integrity test are essential
Seal integrity can be confirmed by conducting following tests
• Tensile test -Placing each side of a one-inch sealed section of the package seal within the grip and pulling one of the seals end away from the other side
• Burst testing-Filling a package by air or gas at a controlled rate, and maintaining the pressure for a specific period and inspecting the failure

• Dye penetrant test-It is a non-destructive test used to dictate surface break flows that is to locate surface crack, porosity, laps, and other surface discontinuity
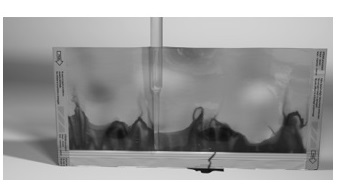
• Ease of opening-It is the test to confirm the seal quality by opening the package and visually observing seal quality
Conclusion
There are roughly 53 million outpatient surgical operations and 46 million inpatient surgical procedures performed in the United States each year. Every year, at least 10 million gastrointestinal endoscopies are performed. Each of these procedures includes contacting a patient's sterile tissue or mucous membranes with a medical device or surgical tool. All of these operations have a significant risk of infection. Failure to adequately disinfect or sterilize equipment poses not just the risk of breaching host barriers, but also the risk of person-to-person transmission and environmental pathogen transmission.
In order to ensure that medical and surgical tools do not transfer infectious organisms to patients, disinfection and sterilization must be achieved using disinfectants and sterilization methods.
References
https://www.iso.org/standard/56137.html
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7099662/
https://www.fda.gov/sterilization-process-controls
https://docbo.com/importance-of-sterilization/